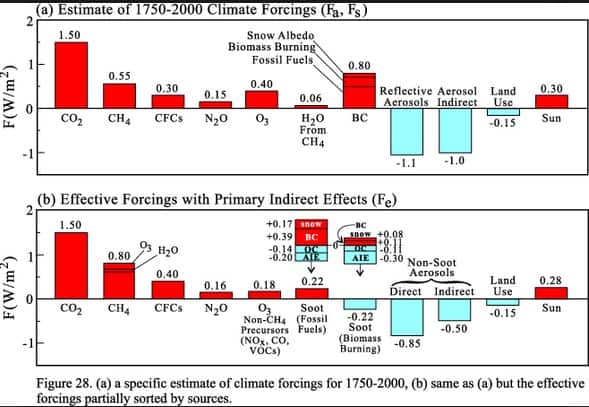
A number of basic factors affect the behaviour of different greenhouse gases as forcing agents within the climate system (Shine et al., 1990). First, the absorption strength and wavelength of the absorption in the thermal infrared are of fundamental importance in dictating whether a molecule can be an important greenhouse forcing agent; this effect is modified by the overlap between the absorption bands and those of other gases present in the atmosphere. For example, the natural quantities of CO2 are so large (compared to other trace gases) that the atmosphere is very opaque over short distance at the centre of its 15m absorption band. The addition of a small amount of gas capable of absorbing at this wavelength has negligible effect on the net radiative flux at the tropopause. Other greenhouse gases with absorption bands in the more transparent regions of the infrared spectrum, in particular between 10 and 12m, will have a far greater radiative forcing effect.
Second, the atmospheric residence time (lifetime) of a greenhouse gas can greatly influence its potential as a radiative forcing agent. Gases that remain in the atmosphere for considerable periods of time, before being removed via their sinks, will have a greater forcing potential over longer time horizons.
Third, the existing quantities of a greenhouse gas in the atmosphere can dictate the effect that additional molecules of that gas can have. For gases such as halocarbons, where the naturally occurring concentrations are zero or very small, their forcing is close to linear for present-day concentrations. Gases such as methane and nitrous oxide are present in such quantities that significant absorption is already occurring, and it is found that their forcing is approximately proportional to the square root of their concentration. For carbon dioxide, parts of the spectrum are already so opaque that additional molecules are almost ineffective; the forcing is found to be only logarithmic in concentration.
As well as the direct effects on radiative forcing, many greenhouse gases also have indirect radiative effects on the climate through their interactions with atmospheric chemical processes. For example, the oxidation of methane in the atmosphere leads to additional production of CO2. Certain halocarbons such as the CFCs significantly affect the distribution of ozone, another greenhouse gas, in the atmosphere. The hydroxyl (OH) radical, itself not a greenhouse gas, is extremely important in the troposphere as a chemical scavenger. Reactions with OH largely control the atmospheric lifetime, and, therefore, the concentrations of a number of greenhouse gases, in particular methane and many of the halocarbons.


Leave a Reply